
Beneficial aspects of high dose intravenous vitamin C on patients with COVID-19 pneumonia in severe condition: a retrospective case series study
Introduction
Coronavirus disease 2019 (COVID-19), the infectious illness caused by severe acute respiratory coronavirus 2 (SARS-CoV-2) has become a worldwide heath care crisis since December 2019 (1,2). The severity of COVID-19 is classified into mild, moderate, severe and critical type on the guideline made by National Health and Family Planning Commission of the People’s Republic of China (3). The severe patient is mainly characterized with deteriorated respiratory function and rapid progression of radiological lesions, and the critical patient further requires mechanical ventilation (MV), accompanying with shock or multiple organ failure (Table S1). The aged patients and those with pre-existing respiratory, cardiac and diabetic disease appear to be at high risk of rapid development to severe and critical type (4). Various methods of respiratory support are the main treatment for the severe and critical patients, especially in the situation of disease aggravation. The respiratory supports are consisted of high flow oxygen therapy, noninvasive or invasive MV and extracorporeal membrane oxygenation (ECMO). As effective antiviral therapy is still vacant, the mortality of COVID-19 patients who required MV was reported as high as 66% (5). Therefore, it is important to develop an effective rescue therapy for these patients, especially in the situation of disease aggravation.
Vitamin C, one of the key antioxidants of the body, has been highlighted recently in the field of critical care medicine (6). High does intravenous vitamin C (HDIVC), a new developed method of administration, has been proven to exert beneficial effect in various critical illness by animal and clinical study (7,8). The rationale for HDIVC application in the treatment of severe and critical COVID-19 patients, as we deemed, relies on (I) its effective ability for eliminating the burst of reactive oxygenase species (ROS) and the following cytokine storm (9), which are the basis for acute respiratory distress syndrome (ARDS). The recent CITRIS-ALI study (10) showed HDIVC (50 mg/kg actual body weight every 6 hours for 96 hours) reduced 28 day all-cause mortality (29.8% vs. 46.3%) in a cohort of septic patients with ARDS. (II) Its immune enhancing property and the potential anti-virus ability (11), which might help for improving lymphopenia, the main characteristic of COVID-19 which was related with severity (12). Also, application of HDIVC in COVID-19 has already got concerns of some scholars (13). Still, promotion of HDIVC needs further evidence.
In this retrospective case series study, we made an observational comparison of outcomes before and after HDIVC in a cohort of severe and critical patients after disease aggravation occurred. The dosage of vitamin C [median (IQR)] applied in severe patients was 162.7 (71.1–328.6) mg/kg (body weight)/day and 178.6 (133.3–350.6) mg/kg/day in critical patients. The main outcomes were mainly divided into inflammatory response, immune and organ dysfunction. To the best of our knowledge, it is the first time to describe the effect of HDIVC on COVID-19 pneumonia. We present the following article according to the STROBE reporting checklist (available at http://dx.doi.org/10.21037/apm-20-1387).
Methods
Study design and participants
This study is a retrospective case series study. It was conducted in accordance with the amended Declaration of Helsinki (as revised in 2013) and was approved by the institutional ethics board of Ruijin Hospital, Shanghai Jiaotong University school of medicine. Oral consents were obtained from each enrolled patient. COVID-19 patients admitted in Shanghai public health clinical center from January 22, 2020 to April 11, 2020 were retrospectively scrolled. The inclusion criteria were: (I) age >18 years; (II) patients who was diagnosed with severe or critical COVID-19 pneumonia. Diagnosis and severity classification were based on the guideline made by National Health and Family Planning Commission of the People’s Republic of China (3) (the specific criteria of severity was shown in Table S1). (III) The severe and critical patients suffered with disease aggravation which was defined as continuous decreasing of PaO2/FiO2 or enlargement of lung radiological lesion for 1–3 days. (IV) The enrolled patients should accept HDIVC within 24 hours after disease aggravation. The judgement of aggravation occurrence and the initiation of HDIVC was performed by Shanghai COVID-19 pneumonia expert group; the dosage of vitamin C was determined according to the common opinion of expert group based on the previous clinical study and research work. (V) patients without being pregnant; (VI) patients without malignant tumor.
Data collection
All the data were collected from electronic medical forms by the same trained physician team. The general information including age, gender, body weight and co-existing diseases were collected. The interval from first symptom to admission and initiation of HDIVC were calculated. The dosage, duration and contemporary maximum oxygen support and other medication (including antibiotics, glucocorticoid and low molecular heparin) were collected. The worst value of outcomes on the day before HDIVC (named as day 0), 3‒5 (named as day 3) and 7‒10 (named as day 7) days after HDIVC were collected. As the intensive monitoring performed immediately after aggravation occurred, all the data of day 0 could be obtained. Most data of days 3 and 7 could also be obtained since the formulated clinical routine test. Outcomes included inflammatory markers [serum level of C-reactive protein (CRP), body temperature, erythrocyte sedimentation rate (ESR)], immune function indicators (counts of lymphocyte and CD4+ T cell), organ function indicators [PaO2/FiO2, D-Dimer, platelet counts, sequential organ failure assessment (SOFA) score, serum level of lactate dehydrogenase (LDH), total bilirubin and creatinine].
Statistical analysis
Continuous variables were presented as medians with interquartile ranges (IQRs) and compared using Mann-Whitney U test or reported as mean with standard deviation and compared with t-test. Categorical variables were presented as frequency with percentage and compared with Fisher’s exact test. Generalized estimating equation (GEE) approach was applied to investigate the effect of HDIVC on inflammatory markers, immune function, and organ function longitudinally over time, adjusted for severity type. All statistical analyses were performed using SAS v. 9.2 (SAS Institute Inc., USA). Two-sided P values of less than 0.05 were deemed to be statistically significant.
Results
Patients characteristics
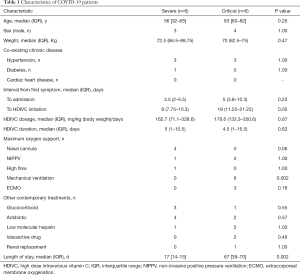
A total of twelve COVID-19 patients were enrolled, among which six of them were classified into severe type, and other six critical type (Figure S1). As Table 1 showed, the median age of severe and critical type was 56 years (IQR, 32–65 years) and 63 years (IQR, 60–82 years) respectively. Three of six were men among severe patients and four of six among critical patients. For severe patients, the median interval from first symptom to admission and HDIVC initiation was 3.5 days (IQR, 2–5.5 days) and 8 days (IQR, 7.8–10.3 days), and for critical patients, the interval was 5 days (IQR, 3.8–10.3 days) and 19 days (IQR, 11.25–21.25 days). Between severe and critical patients, no statistically significant differences were found regarding age (P=0.26), body weight (P=0.47) and interval from onset to admission (P=0.23). The interval from disease onset to HDIVC initiation was longer in critical than severe patients with borderline significance (P=0.05). The duration and dosage of HDIVC showed no evidence of difference between two types (P>0.05). The dosage of vitamin C [median (IQR)] applied in severe patients was 162.7 (71.1–328.6) mg/kg (body weight)/day and 178.6 (133.3–350.6) mg/kg/day in critical patients. Among severe patients, four cases were treated with oxygen therapy of nasal cannula with 3 litters/L, one case with high flow oxygen therapy and one case with non-invasive positive pressure ventilation. All critical patients were treated with mechanical ventilation (MV). Among them, three cases also accepted ECMO, two cases received vasoactive drug due to circulatory failure, one case received continuous renal replacement therapy. Other contemporary treatments accompanying with HDIVC after disease aggravation included glucocorticoid, antibiotics, low molecular heparin (Table 1). The length of stay was significantly longer in critical [67 (IQR, 39–70) days] than severe patients [17 (IQR, 14–15) days; P=0.02].
Full table
HDIVC on inflammatory response
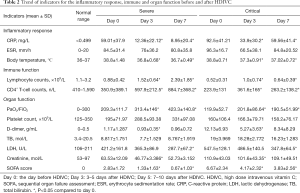
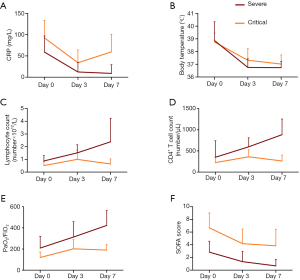
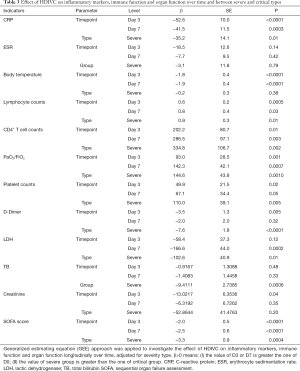
Elevation of serum CRP (Table 2) level was observed in both severe (59.01±37.9 mg/L) and critical (92.5±41.21 mg/L) patients as aggravation occurred, which then decreased continuously (Figure 1A) at day 3 (12.36±22.12 mg/L) and day 7 (8.95±20.4 mg/L) in severe patients. For critical patients, CRP decreased at day 3 (33.9±30.2 mg/L) and then slightly increased at day 7 (59.56±41.4 mg/L). The body temperature (Table 2) which partly reflects inflammatory response decreased continuously to almost normal level along with time after HDIVC in both cohorts (Figure 1B). However, the ESR did not show decreasing trend after HDIVC. GEE models (Table 3) further proved the downregulating effect of HDIVC on CRP at day 3 (P<0.0001) and day 7 (P=0.003) days as well as the body temperature at day 3 (P<0.0001) and day 7 (P<0.0001) days in both cohorts. The ameliorating effect of HDIVC on CRP in severe patients was statistically better than the one in critical patients (P=0.0125).
Full table
Full table
HDIVC on immune function
On aggravation, lymphocytopenia (Table 2) occurred in both severe (0.88±0.42, ×109/L) and critical (0.52±0.31, ×109/L) patients, and its subgroup CD4+ T-cell (Table 2) showed the same trend (severe: 350.9±389.1, number/L; critical: 223.9±131, number/L). At days 3 and 7, the lymphocyte (1.52±0.64, 2.39±1.85, ×109/L, Figure 1C) and CD4+ T-cell (597.9±212.5, 884.7±368.2, number/l, Figure 1D) counts renormalized in severe patients. For critical patients, the lymphocytopenia remained after HDIVC, and CD4+ cell counts increased at day 3 with following decreased at day 7. The GEE models (Table 3) showed the general increasing trend of lymphocyte and CD4+ T cell counts at days 3 and 7 after HDIVC (all P<0.05). The immune function enhancing profile were more marked in severe group compared to critical group after HDIVC (P=0.0109 for lymphocyte, P=0.0017 for CD4+ T cell).
HDIVC on organ functions
PaO2/FiO2 (Table 2), representing the respiratory function, decreased after aggravation (209.3±111.7) and returned to normal level at day 3 (313.4±146) and day 7 (423.3±140.8) in severe patients (Figure 1E). For critical patients, PaO2/FiO2 increased from day 0 (119.9±52.7) to day 3 (201.8±86.64) and day 7 (190.5±51.99). By GEE model (Table 3), the HDIVC related improving effect was proved (P=0.0011 for day 3, 0.0007 for day 7). The difference of HDIVC improving effect between severe and critical type was significant (P=0.001). D-Dimer (Table 2) representing coagulation function slightly increased in severe group (1.17±1.287, mg/mL) when aggravation occurred but with a greater extent in critical group (12.13±6.93, mg/mL). After HDIVC treatment, D-Dimer in critical patient decreased markedly at day 3 but not at day 7 in both groups. In GEE model (Table 3), significant difference was found in decreasing of D-Dimer between day 3 (not day 7) versus day 0. The improving effect of HDIVC on D-Dimer was better in critical than severe group (P<0.0001). PLT (Table 2) counts stayed in normal range during the observational period. The liver function indicator LDH (Table 2) level increased abnormally as aggravation occurred in severe (421.2±161.8 U/L) and critical (547.5±128.1 U/L) group, and showed decreasing trend in both groups. GEE model showed significant decrease of LDH only occurred at day 7 (P=0.0002) but not day 3 as compared to day 0. The improving effect of HDIVC on LDH was better in critical than severe group (P=0.0122). Another liver function total bilirubin (Table 2) was among the normal range after HDIVC. Accordingly, the SOFA score (Table 2), contributed mainly by respiratory dysfunction in severe and critical patients, showed the same decreasing trend as PaO2/FiO2 did at days 3 and 7 after HDIVC (Figure 1F). SOFA score (Table 3) improved better in severe group than critical group by HDIVC as GEE model showed (P=0.0004).
Discussion
In this retrospective case series study, we mainly focused on the effect of HIDVC on COVID-19 in three aspects: inflammatory response, immune function and organ function. We showed improvement of CRP, body temperature, lymphocyte counts, CD4+ T cell counts, PaO2/FiO2 and SOFA score within ten days after HDIVC in two cohorts of severe and critical patients (Figure S2). We also found the improving effect of HDIVC on the outcomes of severe patients was better than the one of critical patients.
In critical ill patients, serum level of vitamin C is reported to drop to 10–30 mol/L (normal level of 80–100 mol/L). This might be related to rapid exhaustion by ROS, dilution due to rapid fluid resuscitation, insufficiency supplement and elimination by renal replacement therapy (14). Vitamin C deficiency was reported correlated with the occurrence of multiple organ failure in critically ill patients (15), and requirement for vitamin C increase with diseases severity (16). As genetically loss of the ability to synthesize vitamin C, human must obtain exogenous vitamin C. Intravenous administration of vitamin C showed excellent supplementary effect by quickly increasing the serum level from mol/L to mmol/L (17). Therefore, it is rational to apply HDIVC in the rescue therapy of COVID-19 in severe condition. Recently, treatment with HDIVC for severe acute respiratory distress syndrome (ARDS) from COVID-19 was recommended (18).
The elevation of serum CRP was observed in both severe and critical groups which were consistent with other studies describing the characteristic of COVID-19 patients in severe situation (12,19). The kinetics of CRP is usually used to track and monitor the inflammatory response caused by infection as its short half-life of 19 hours (20). CRP level was shown with rapid reduction by HDIVC (200 mg/kg/day) in a previous before after study in a cohort of sepsis patient (21). Similarly, we showed CRP decreased significantly within ten days after HDIVC by building GEE models. Combined with the trend of body temperature, we deemed that HDIVC might be beneficial for the inflammatory response in COVID-19 patients.
Lymphocytopenia is main characteristic of COVID-19 patients. Recently, the reduction of lymphocyte subset including CD4+, CD8+ and CD3+ T cell was reported to be correlated with COVID-19 severity (22). The underlying mechanism is complex, mainly contributed to the invasion of virus itself. SARS-CoV-2 might causes growth inhibition and apoptosis of hematopoiesis by promotion of autoimmune antibody (23). SARS-CoV-2 also leads the apoptosis of T lymphocyte by affected dendritic cell and production of certain cytokine (24). Consistent with these studies, we showed lymphocyte and its subgroup CD4+ T cell was in subnormal level as aggravation occurred. Lymphocytes, especially T lymphocyte, have been extensively studied in the context of vitamin C biology (25). Both in vitro and in vivo studies showed vitamin C was essential for the development, maturation and proliferation of functional T-lymphocytes and epigenetic regulation of gene expression is one of the underlying mechanisms (26). In our study, we showed lymphocyte and its subgroup CD4+ T cell returned to normal level in severe patients and increased markedly in critical patients after HDIVC treatment. Our findings partly supported the immune enhancing profile of HDIVC in the treatment of COVID-19.
Multiple organs failure was reported in COVID-19 patients with severe situation (12). The most notable involved organ is the lung. The disease aggravation often started with a rapid development of respiratory failure and decreased PaO2/FiO2. Other organ systems secondary to lung include coagulation system, kidney, liver and heart. Decreased platelet counts and elevation of D-D dimer, serum of creatinine, lactic dehydrogenase, glutamic oxaloacetic transaminase, cardiac troponin and creatinine kinase were usually found in severe and critical patients (12). This phenomenon was also partly shown by our study. The organ protection by HDIVC was proved by preclinical and clinical studies in various models of critical condition (27,28). The mechanism relies on the rapid scavenging of ROS and attenuation of ROS related endothelial dysfunction which cause multiple organ failure, especially the ARDS. Other specific mechanisms of HDIVC preventing ARDS include the regulatory of NETosis (29), reducing inflammation via attenuation of NF-κB activation (30), enhancing lung epithelial barrier function by promoting epigenetic and transcriptional expression of protein channels at the alveolar capillary membrane which help regulate alveolar fluid clearance (31). In our study, PaO2/FiO2 were significantly improved after HDIVC in both severe and critical patients. It is noted that the superior respiratory support, such as MV or ECMO, had been already applied before the initiation of HDIVC, therefore the increase of PaO2/FiO2 was probably contributed to usage of HDIVC but not the enhancement of respiratory support. SOFA score also showed decreasing trend after HDIVC, and this result are consistent with Fowler et al.’s early findings (21). However, in their latest prospective study named as CITRIS-ALI trial (10), Fowler et al.’s showed opposite result with much larger sample size. The dosage of vitamin C in their study was 50 mg/kg (body weight) per 6 hours which are similar as ours [162.7 (71.1–328.6) mg/kg/day for severe and 178.6 (133.3–350.6) mg/kg/day for critical patients]. The inconsistence between our study and CITRIS-ALI trial is mainly due to the trial design, and the varies etiologies of enrolled patients and their higher SOFS score (9.8–10.3).
Besides, GEE model showed HDIVC exert better improving effect on the outcomes of severe than the one of critical patients. This might be explained as: (I) the difference of severity between two cohorts, and the severe patients might get better prognosis than critical patients as they received the almost same dosage of vitamin C (Table 1); (II) the interval from admission to HDIVC initiation of severe patients [8 (IQR, 7.8–10.3) days] is shorter than the one of critical patients [19 (IQR, 11.25–21.25) days]. Although without statistically difference (P=0.05), it implies that the earlier application of HDIVC could gain more benefits.
It is reported that high dose, long term, oral application of vitamin C was related with the formation of renal oxalate stone (32) which might cause potential harm to renal function. Therefore, the safety profile of vitamin C should be concerned. The latest case serious study including 157 patients showed no renal stone was reported after temporary, intravenous, high dose application of vitamin C (33). In our study, no patient was observed to suffer any identifiable adverse event related with HDIVC.
Limitation: (I) relative fewer patients accepted HDIVC rescue therapy restrained the study design as the before and after treatment study and small sample size. No controlled group was applied. These defects limit the power of study; (II) the dosage and duration varied relative widely depending on clinical situation and clinician experience. Based on the experiences obtained from these cases and our previous work, we promoted a HDIVC protocol (Figure S3) and its related prospective controlled clinical trial (ChiCTR2000032400) are being conducted now; (III) the interfering effect of concomitant therapies such as glucocorticoid, antibiotic, low molecular heparin could not be excluded. Therefore, HDIVC might be regarded as compassionate similar to Remdesivir reported recently (34); (IV) the regression to the mean is an unavoidable phenomenon, which might overstate the therapeutic effect of HDIVC.
Conclusions
In the preliminary retrospective study, HDIVC might be regarded as an important rescue therapy in aggravation of severe and critical COVID-19 patients according to the observational improvement of inflammatory response, immune function and organ function. The limited sample size and study design preclude a definitive statement about the potential effectiveness of HDIVC, and these observations require evaluation in clinical trials.
Acknowledgments
We thank all the staff of Shanghai public health clinical center for their great effort on the treatment of COVID-19 pneumonia patients.
Funding: The second batch of emergency key scientific and technological project of Shanghai Municipal Committee of Science and Technology (20411950300/20411950301). The Clinical Research Project of Ruijin Hospital Affiliated to Shanghai Jiao Tong University School of Medicine (2018CR004) to Enqiang Mao. National Natural Science Foundation of China (81870311) to Jun Huang.
Footnote
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at http://dx.doi.org/10.21037/apm-20-1387
Data Sharing Statement: Available at http://dx.doi.org/10.21037/apm-20-1387
Peer Review File: Available at http://dx.doi.org/10.21037/apm-20-1387
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/apm-20-1387). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. It was conducted in accordance with the amended Declaration of Helsinki (as revised in 2013) and was approved by the institutional ethics board of Ruijin Hospital, Shanghai Jiaotong University school of medicine. Oral consents were obtained from each enrolled patient.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Mahase E. Covid-19: WHO declares pandemic because of "alarming levels" of spread, severity, and inaction. BMJ 2020;368:m1036. [Crossref] [PubMed]
- Spinelli A, Pellino G. COVID-19 pandemic: perspectives on an unfolding crisis. Br J Surg 2020;107:785-7. [Crossref] [PubMed]
- Guideline for Diagnosis and Treatment of SARS-CoV-2 (the seventh edition). National Health and Family Planning Commission of the People’s Republic of China. 2020. Available online: http://www.nhc.gov.cn/yzygj/s7653p/202003/46c9294a7dfe4cef80dc7f5912eb1989.shtml
- Weiss P, Murdoch DR. Clinical course and mortality risk of severe COVID-19. Lancet 2020;395:1014-5. [Crossref] [PubMed]
- Wu C, Chen X, Cai Y, et al. Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern Med 2020;180:934-43. [Crossref] [PubMed]
- Brant EB, Angus DC. Is High-Dose Vitamin C Beneficial for Patients With Sepsis? JAMA 2019;322:1257-8. [Crossref] [PubMed]
- Oudemans-van Straaten HM, Spoelstra-de Man AM, de Waard MC. Vitamin C revisited. Crit Care 2014;18:460. [Crossref] [PubMed]
- Marik PE, Khangoora V, Rivera R, et al. Hydrocortisone, Vitamin C, and Thiamine for the Treatment of Severe Sepsis and Septic Shock: A Retrospective Before-After Study. Chest 2017;151:1229-38. [Crossref] [PubMed]
- Moore JB, June CH. Cytokine release syndrome in severe COVID-19. Science 2020;368:473-4. [Crossref] [PubMed]
- Fowler AA 3rd, Truwit JD, Hite RD, et al. Effect of Vitamin C Infusion on Organ Failure and Biomarkers of Inflammation and Vascular Injury in Patients With Sepsis and Severe Acute Respiratory Failure: The CITRIS-ALI Randomized Clinical Trial. JAMA 2019;322:1261-70. [Crossref] [PubMed]
- van Gorkom GNY, Klein Wolterink RGJ, Van Elssen CHMJ, et al. Influence of Vitamin C on Lymphocytes: An Overview. Antioxidants (Basel) 2018;7:41. [Crossref] [PubMed]
- Wang D, Hu B, Hu C, et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA 2020;323:1061-9. [Crossref] [PubMed]
- Cheng RZ. Can early and high intravenous dose of vitamin C prevent and treat coronavirus disease 2019 (COVID-19)? Med Drug Discov 2020;5:100028. [Crossref] [PubMed]
- Carr AC, Rosengrave PC, Bayer S, et al. Hypovitaminosis C and vitamin C deficiency in critically ill patients despite recommended enteral and parenteral intakes. Crit Care 2017;21:300. [Crossref] [PubMed]
- Borrelli E, Roux-Lombard P, Grau GE, et al. Plasma concentrations of cytokines, their soluble receptors, and antioxidant vitamins can predict the development of multiple organ failure in patients at risk. Crit Care Med 1996;24:392-7. [Crossref] [PubMed]
- Carr AC, Rosengrave PC, Bayer S, et al. Hypovitaminosis C and vitamin C deficiency in critically ill patients despite recommended enteral and parenteral intakes. Crit Care 2017;21:300. [Crossref] [PubMed]
- de Grooth HJ, Manubulu-Choo WP, Zandvliet AS, et al. Vitamin C Pharmacokinetics in Critically Ill Patients: A Randomized Trial of Four IV Regimens. Chest 2018;153:1368-77. [Crossref] [PubMed]
- Matthay MA, Aldrich JM, Gotts JE. Treatment for severe acute respiratory distress syndrome from COVID-19. Lancet Respir Med 2020;8:433-4. [Crossref] [PubMed]
- Shen C, Wang Z, Zhao F, et al. Treatment of 5 Critically Ill Patients With COVID-19 With Convalescent Plasma. JAMA 2020;323:1582-9. [Crossref] [PubMed]
- Williams AM, Ladva CN, Leon JS, et al. Changes in micronutrient and inflammation serum biomarker concentrations after a norovirus human challenge. Am J Clin Nutr 2019;110:1456-64. [Crossref] [PubMed]
- Fowler AA 3rd, Syed AA, Knowlson S, et al. Phase I safety trial of intravenous ascorbic acid in patients with severe sepsis. J Transl Med 2014;12:32. [Crossref] [PubMed]
- Xu B, Fan CY, Wang AL, et al. Suppressed T cell-mediated immunity in patients with COVID-19: A clinical retrospective study in Wuhan, China. J Infect 2020;81:e51-e60. [Crossref] [PubMed]
- Yang M, Li CK, Li K, et al. Hematological findings in SARS patients and possible mechanisms Int J Mol Med 2004;14:311-5. (review). [Crossref] [PubMed]
- Channappanavar R, Zhao J, Perlman S. T cell-mediated immune response to respiratory coronaviruses. Immunol Res 2014;59:118-28. [Crossref] [PubMed]
- van Gorkom GNY, Klein Wolterink RGJ, Van Elssen CHMJ, et al. Influence of Vitamin C on Lymphocytes: An Overview. Antioxidants (Basel) 2018;7:41. [Crossref] [PubMed]
- Manning J, Mitchell B, Appadurai DA, et al. Vitamin C promotes maturation of T-cells. Antioxid Redox Signal 2013;19:2054-67. [Crossref] [PubMed]
- Spoelstra-de Man AME, Elbers PWG, Oudemans-van Straaten HM. Making sense of early high-dose intravenous vitamin C in ischemia/reperfusion injury. Crit Care 2018;22:70. [Crossref] [PubMed]
- Marik PE. Vitamin C for the treatment of sepsis: The scientific rationale. Pharmacol Ther 2018;189:63-70. [Crossref] [PubMed]
- Mohammed BM, Fisher BJ, Kraskauskas D, et al. Vitamin C: a novel regulator of neutrophil extracellular trap formation. Nutrients 2013;5:3131-51. [Crossref] [PubMed]
- Chen Y, Luo G, Yuan J, et al. Vitamin C mitigates oxidative stress and tumor necrosis factor-alpha in severe community-acquired pneumonia and LPS-induced macrophages. Mediators Inflamm 2014;2014:426740. [Crossref] [PubMed]
- Fisher BJ, Kraskauskas D, Martin EJ, et al. Mechanisms of attenuation of abdominal sepsis induced acute lung injury by ascorbic acid. Am J Physiol Lung Cell Mol Physiol 2012;303:L20-32. [Crossref] [PubMed]
- Chen X, Shen L, Gu X, et al. High-dose supplementation with vitamin C--induced pediatric urolithiasis: the first case report in a child and literature review. Urology 2014;84:922-4. [Crossref] [PubMed]
- Prier M, Carr AC, Baillie N. No Reported Renal Stones with Intravenous Vitamin C Administration: A Prospective Case Series Study. Antioxidants (Basel) 2018;7:68. [Crossref] [PubMed]
- Grein J, Ohmagari N, Shin D, et al. Compassionate Use of Remdesivir for Patients with Severe Covid-19. N Engl J Med 2020;382:2327-36. [Crossref] [PubMed]


